Medical Devices Testing and Evaluation Centre (MD-TEC)
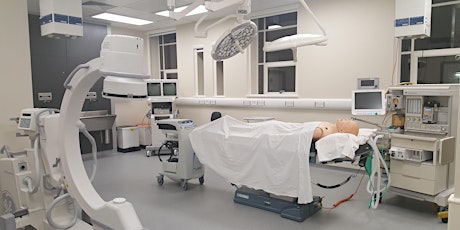
Medical Devices Testing and Evaluation Centre (MD-TEC) is a state of the art facility based within the Institute of Translational Medicine in Birmingham, designed to support medical technology and life science businesses across the region.
Combining unrivalled facilities with clinical, academic and industry expertise, MD-TEC is designed to remove the regulatory blockages encountered by SMEs and to support the accelerated translation of medical devices in to the commercial environment.
Medical Devices Testing and Evaluation Centre (MD-TEC)
Upcoming (0)
Sorry, there are no upcoming events
Past (10)

MD-TEC Workshop: Navigating the MDR (Medical Device Regulations) Routes to CE marking, 07/06/2018
Thu, Jun 7, 9:00 AM
Free

MD-TEC Workshop: Navigating the MDR (Medical Device Regulations) Routes to CE marking, 24/04/2018
Tue, Apr 24, 1:00 PM
Free

MD-TEC Workshop: The Role of Human Factors Engineering for Development of Medical Devices, 10/04/2018
Tue, Apr 10, 1:00 PM
Free

MD-TEC Workshop: Device Testing Using Organotypic Culture Systems, 15/03/2018
Thu, Mar 15, 1:00 PM
Free

MD-TEC Workshop: Drug & Device Discovery and Optimisation for Clinical Development, 15/03/2018
Thu, Mar 15, 8:30 AM
Free